Compost Seed of Trichoderma harzianum UD12-102 in Controlling Collar and Stem Rot of Tomato Caused by Sclerotium rolfsii
Main Article Content
Abstract
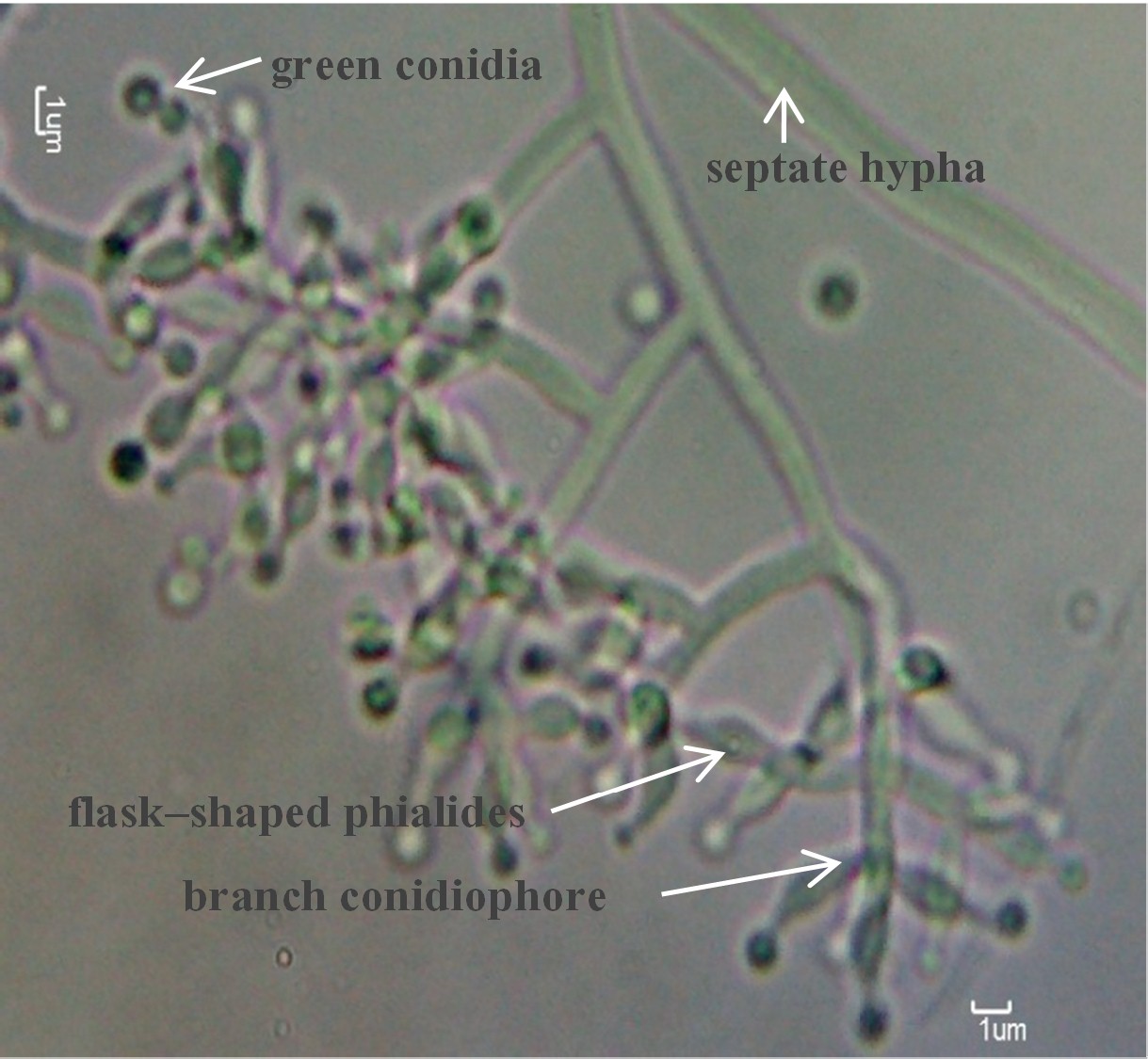
Antagonistic fungus, Trichoderma harzianum UD12-102, exhibited 90% inhibition against Sclerotium rolfsii in vitro and 80% survival of tomatoes infected by S. rolfsii in vivo. Moreover, the antagonistic fungi increased the effectiveness of a commercial fungicide (vitavax) in controlling S. rolfsii in tomatoes. In field experiments, composts were used as carriers for T. harzianum UD12-102 inoculum preparation. Following S. rolfsii inoculation, the survival percentages of tomato plants were not significantly different with all treatments receiving T. harzianum UD12-102 antagonist. However, the inoculum prepared with compost B (inoculated the antagonist at beginning of composting) resulted in a high survival percentage (more than 60%) with 4 weeks of infection, while the survival percentage of control plants dramatically decreased on week 2 (8.35%), and the plants died after 3 weeks due to S. rolfsii. The compost was a good alternative carrier for antagonistic fungi inoculation and was friendly with soil environments.
Article Details
Published articles are under the copyright of the Environment and Natural Resources Journal effective when the article is accepted for publication thus granting Environment and Natural Resources Journal all rights for the work so that both parties may be protected from the consequences of unauthorized use. Partially or totally publication of an article elsewhere is possible only after the consent from the editors.
References
Asran-Amal A, Moustafa-Mahmoud SM, Sabet KK, El-Banna OH. In vitro antagonism of cotton seedlings fungi and characterization of chitinase isozyme activities in Trichoderma harzianum. Saudi Journal of Biological Sciences 2010;17:153-7.
Danon M, Chen Y, Hadar Y. Ascomycete communities associated with suppression of Sclerotium rolfsii in compost. Fungal Ecology 2010;3:20-30.
Dongarmart P, Saksirirat W, Sirimungkararat S, Chanthabut R, Saepaisan S. Selection of a suitable substrate for biomass production of antagonistic fungi, Trichoderma harzianum (T9) and Trichoderma asperellum (T13). Khon Kaen Agriculture Journal 2015;43(supplement 1):922-7.
El-Mohamedy RSR, Jabnoun-Khiareddine H, Daami-Remadi M. Control of root rot diseases of tomato plants caused by Fusarium solani, Rhizoctonia solani and Sclerotium rolfsii using different chemical plant resistance inducers. Tunisian Journal of Plant Protection 2014;9:1.
Fahima T, Henis Y. Interaction between pathogen, host and biocontrol agent: multiplication of Trichoderma hamatum and Talaromyces flavus on roots of diseased and healthy hosts. In: Hornby D, editor. Biological Control of Soilborne Plant Pathogens. London: CAB International; 1990. p. 165-80.
Ganesan S, Kuppusamy RG, Sekar R. Integrated management of stem rot disease (Sclerotium rolfsii) of groundnut (Arachis hypogaea L.) using Rhizobium and Trichoderma harzianum (ITCC - 4572). Turkish Journal of Agriculture and Forestry 2007;31:103-8.
Ganeshan G. Fungicidal control of Sclerotium basal rot of cluster bean cv. Pusa Naubahar. Indian Phytopathology 1997;50:508-12.
Harman GE, Kubicek CP. Trichoderma and Gliocladium. CRC Press; 2002.
Jomduang J, Sariah M. Antagonistic effect of Malaysian isolates of Trichoderma harzianum and Gliocladium virens on Sclerotium rolfsii. Pertanika Journal of Tropical Agricultural Science 1997;20:35-41.
Kulkarni S. Chemical control of foot rot of wheat in Karnataka. Pesticides 1980;14:29-30.
Libardi N, Soccol CR, Góes-Neto A, de Oliveira J, Vandenberghe LPS. Domestic wastewater as substrate for cellulase production by Trichoderma harzianum. Process Biochemistry 2017;57:190-9.
Manu TG, Nagaraja A, Chetan SJ, Vinayaka H. Efficacy of fungicides and biocontrol agents against Sclerotium rolfsii causing foot rot disease of finger millet, under in vitro conditions. Global Journal of Biology, Agriculture and Health Sciences 2012;1:46-50.
Okereke VC, Wokocha RC, Godwin-Egein MI. Evaluation of Trichoderma harzianum, some botanicals and a fungicide on sclerotium wilt of potted tomato. Agricultural Journal 2007;2:453-6.
Pan S, Sen C. Chemical control of foot rot of wheat caused by Sclerotium rolfsii Sacc. Proceedings of the Indian National Science Academy. Part B: Biological Sciences 1997;43:79-85.
Patil MS, Patil A. The rust fungi: systematics, diseases and their management. In: Arya A, Perello AE, editors. Management of Fungal Plant Pathogens. London: CAB International; 2010. p. 201-16.
Rahman ML, Haque MS, Muqit A, Alam KB, Ali S. Response of Phytophthora parasitica to different fungicides. Bangladesh Journal of Plant Pathology 1994;10:35-6.
Saravanakumar K, Fan L, Fu K, Yu C, Wang M, Xia H, Sun J, Li Y, Chen J. Cellulase from Trichoderma harzianum interacts with roots and triggers induced systemic resistance to foliar disease in maize. Scientific Reports 2016;6:35543.
Schubert M, Mourad S, Fink S, Schwarze FWMR. Ecophysiological responses of the biocontrol agent Trichoderma atroviride (T-15603.1) to combined environmental parameters. Biological Control 2009;49:84-90.
Thiribhuvanamala G, Rajeswari E, DuraiSwamy S. Biological control of stem rot of tomato caused by Sclerotium rolfsii Sacc. Madras Agricultural Journal 1999;86:30-3.
Thrane C, Tronsmo A, Jensen DF. Endo-1,3-β-glucanase and cellulase from Trichoderma harzianum: purification and partial characterization, induction of and biological activity against plant pathogenic Pythium spp. European Journal of Plant Pathology 1997;103:331-44.
Tsahouridou PC, Thanassoulopoulos CC. Proliferation of Trichoderma koningii in the tomato rhizosphere and the suppression of damping-off by Sclerotium rolfsii. Soil Biology and Biochemistry 2002;34:767-76.
Wijesinghe CJ, Wijeratnam RSW, Samarasekara JKRR, Wijesundera RLC. Biological control of Thielaviopsis paradoxa on pineapple by an isolate of Trichoderma asperellum. Biological Control 2010;53:285-90.
Zmora-Nahum S, Danon M, Hadar Y, Chen Y. Chemical properties of compost extracts inhibitory to germination of Sclerotium rolfsii. Soil Biology and Biochemistry 2008;40:2523-9.